The Medicines and Healthcare Products Regulatory Agency (MHRA) in the United Kingdom announced the launch of its comprehensive Public Assessment Report Database (PARD). With PARD, individuals, healthcare professionals, and researchers can now easily access a wealth of information about medicinal products, fostering transparency, informed decision-making,…
News
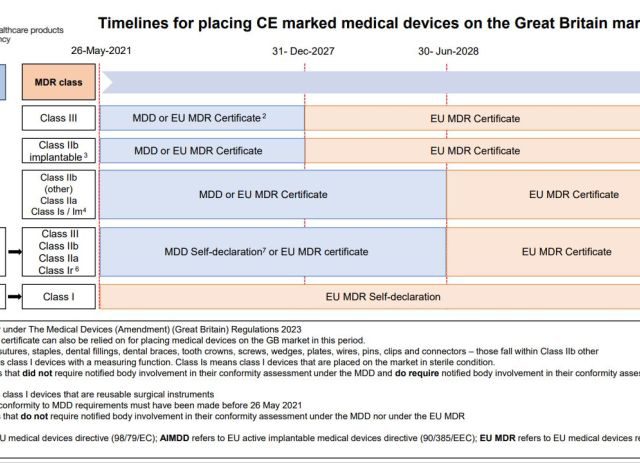
The MHRA has Published a Statutory Instrument
The MHRA has Published a Statutory Instrument to Extend Timelines for Certain Devices Compliant with EU Legislation They have also given a useful infographic setting out the dates for IVDs and Medical Devices. https://lnkd.in/ef4v6Pfd
Medicines and Healthcare products
The Medicines and Healthcare products Regulatory Agency (MHRA), in conjunction with the National Institute for Health and Care Excellence (NICE), Health Technology Wales (HTW) and Scottish Health Technology Group (SHTG) will initiate a new regulatory pathway set to support safe patient access to innovative medical…

Office for Product Safety and Standard’s
The Office for Product Safety and Standard’s new UKMCAB service is now LIVE. You can access the service by visiting the following new web address. https://lnkd.in/ey2Z4xK3

Virtual manufacturing of medical devices
On 17 May 2023 The MHRA Updated its Guidance Virtual manufacturing of medical devices. It has some useful information about the documentation required and the responsibilities. https://lnkd.in/d6sHDTM

Video of the Features and Benefits
Check out this overview video of the features and benefits of the free-to-use, publicly accessible online database, UKAS CertCheck. Register for a free account and search accredited management system certifications here:

World Athletics (WA)
📣 World Athletics (WA) and the World Federation of the Sporting Goods Industry (WFSGI) have signed a Memorandum of Understanding (MoU) to continue and strengthen their collaboration. 🎉 Emma P. (Mason) Zwiebler, Robbert De Kock, Jon Ridgeon, Sylvia Barlag Read the full Press Release here…

MDCG 2022-18
MDCG 2022-18 – MDCG Position Paper on the application of Article 97 MDR to legacy devices for which the MDD or AIMDD certificate expires before the issuance of an MDR certificate. As I am sure you are already aware, the new Article 97 basically allows…

SwissMedic has raised a new FAQ
SwissMedic has raised a new FAQ document regarding the registration of Medical Devices on the Swiss Market. Some of the answers may surprise you! Download a copy here:- file:///C:/Users/Martin/AppData/Local/Temp/MicrosoftEdgeDownloads/e5949b5e-8c43-42a3-a550-46d2320cfd21/BW630_30_009e_MB_FAQ_Notifications.pdf
Did you know ISO/TR 24971
Did you know ISO/TR 24971, Medical devices – Guidance on the application of ISO 14971, is a companion document to the globally recognized risk management standard ISO 14971, Medical devices – Application of risk management to medical devices. The ISO standard, which was updated in…




